Automated stent defect detection and classification with a high numerical aperture optical system
Carlos has collaborated since 2010 on the development of confocal, interferometry and focus variation technologies at Sensofar, where he holds the R&D Engineering Manager position since 2018. His interests are Optomechanical Systems Design and Image Processing.
Its consolidated research work in optical engineering confer the Sensofar R&D group an outstanding position to always stay up-to-date in terms of innovation and the highest technological level.
Automated stent defect detection and classification with a high numerical aperture optical system full article
C. Bermudez,1,2 F. Laguarta,1,3 C. Cadevall,1,2 A. Matilla,2 S. Ibañez,1 R. Artigas1,2
1Universitat Politècnica de Catalunya (UPC) Rambla Sant Nebridi, 10, E-08222 Terrassa, Spain.
2Sensofar-Tech, S.L., (Spain)
3Sensofar-Medical, S.L., (Spain)
Proceedings Volume 10334, Automated Visual Inspection and Machine Vision II; 103340C (2017)
Event: SPIE Optical Metrology, 2017, Munich, Germany
Abstract
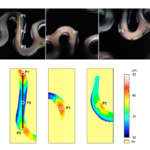
Stent quality control is a highly critical process. Cardiovascular stents have to be inspected 100% so as no defective stent is implanted in a human body. However, this visual control is currently performed manually and every stent could need tenths of minutes to be inspected. In this paper, a novel optical inspection system is presented. By the combination of a high numerical aperture (NA) optical system, a rotational stage and a line-scan camera, unrolled sections of the outer and inner surfaces of the stent are obtained and image-processed at high speed. Defects appearing in those surfaces and also in the edges are extremely contrasted due to the shadowing effect of the high NA illumination and acquisition approach. Therefore by means of morphological operations and a sensitivity parameter, defects are detected. Based on a trained defect library, a binary classifier sorts each kind of defect through a set of scoring vectors, providing the quality operator with all the required information to finally take a decision. We expect this new approach to make defect detection completely objective and to dramatically reduce the time and cost of stent quality control stage.