Measurement modes
Imaging

Screenshot

Unrolled FOV

Unrolled section
3D Modes

Surface topography

Surface roughness

Coating thickness
Inspection capabilities

Outer surface

Inner surface
Lateral surface

Edges
Measurable stent
| Stent types | Metallic, color coated, polymer, welded, with grooves and braided |
| OD | 1 – 32 mm |
| Length | Up to 200mm |
Light sources
| Flexible illumination | Up to 9 light sources available |
| Standard illumination | TOP white light, BACK green light and two SIDE white lights |
Dimensions

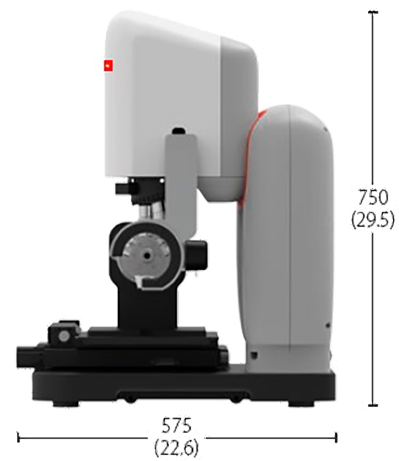
Sensorhead & Controller
Objectives performance
| MAG | 2X | 5X | 10X |
| Numerical Aperture | 0.055 | 0.14 | 0.28 |
| Working Distance (mm) | 34 | 34 | 34 |
| Horizontal FOV (mm) | 9 | 3.6 | 1.8 |
| Spatial sampling (µm)[1] | 4.4 | 1.76 | 0.88 |
| Optical resolution (µm)[2] | 2.77 | 1.09 | 0.54 |
| Unrolled acquisition rate (mm2/s)[3] | 120 | 20 |
1 Pixel size on the imaged surface. 2 L&S: Line and Space (500nm wavelength)
3 Frame rate 3000 fps.
| MAG | 10XDI | 20XDI | 50XDI |
| Numerical Aperture | 0.3 | 0.4 | 0.55 |
| Working Distance (mm) | 7.4 | 4.7 | 3.4 |
| Horizontal FOV (mm) | 1.8 | 0.9 | 0.36 |
| Spatial sampling (µm)[1] | 0.88 | 0.44 | 0.18 |
| Optical resolution (µm)[2] | 0.51 | 0.38 | 0.28 |
| Vertical resolution (nm)[4] | 1 | 1 | 1 |
1 Pixel size on the imaged surface. 2 L&S: Line and Space (500nm wavelength)
4 Vibration isolation table is required.
Computer & Operating System

| Computer | HP Z220 SFF, HP display 2560×1440 pixels |
| Computer Dimensions (WxDxH) | 338x381x100 mm |
| Monitor Dimensions (WxDxH) | 614x218x534 mm (with stand) |
| Operating System | Windows 10, 64 bits |
Data management
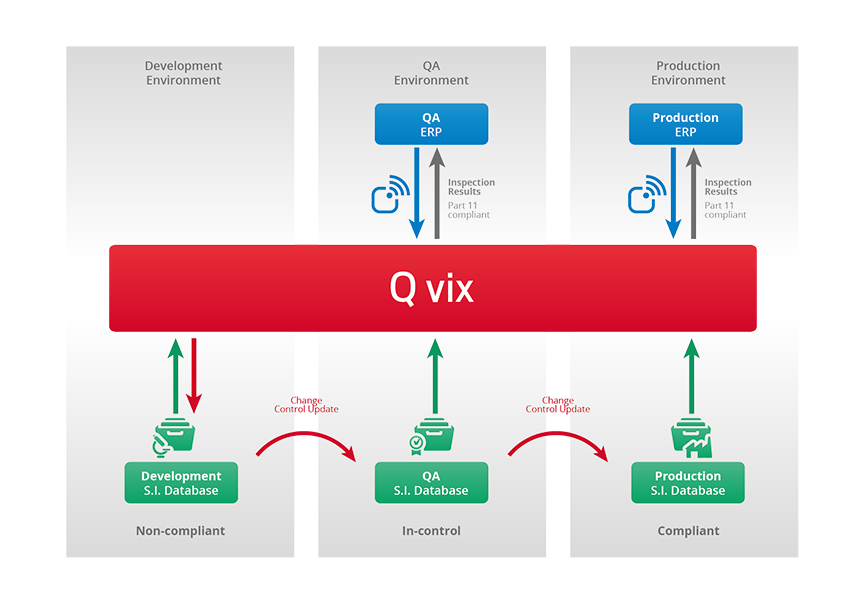
Depending on the intended use, the Q vix can be operated in three different environments: Development, Quality Assurance (QA) and Production. Each environment has its own databases.
Data is organized in three different categories:
1 System Databases containing User data and System configuration files
2 Stent Inspection Databases containing Stent Model data, Defect libraries and Acquisition, Analysis and Report recipes
3 Inspection Results

530nm



