CORONARY STENTS
Coronary stents are the most common type of stents and the primary and most extended inspection application for both Q six and Q vix, which perform the measurement of coronary stents.
With sizes ranging from 1mm to 3mm and lengths ranging from 8mm to 38mm, coronary stents require a high throughput in the inspection. The simultaneous dimensional and visual analysis provided by Sensofar Medical reduces inspection to a single step, increasing the efficiency of the overall process and reducing inspection cost.

Adaptable inspection for any stent material and design
Powerful software tools allow the inspection of any type of coronary stent, adapting the analysis to a variety of stent typologies and materials, from standard metallic stents to polymer stents and including devices with non-metallic finishing or with welded structures.


Simultaneous dimensional analysis and visual inspection
The hardware and software of Sensofar Medical’s inspection systems have been designed to perfectly accommodate a stent’s cylindrical geometry, offering a unique combination of automatic dimensional measurements and visual inspection that dramatically increases inspection throughput to keep up with production requirements.
As an example, Q six can achieve an inspection time as low as 2 minutes on the complete inspection of a 1.5mm OD and 33mm length, including dimensional measurement on hundreds of positions and automatic visual inspection in the outer and inner surfaces.
Automatic inspection of the stent’s inner surface
The optical design of Sensofar Medical’s inspection systems, and our unique mandrels, allow the inspection of the inner surface of the stent.
The high quality images obtained from the inner surface of the stent enable automatic visual inspection of the stent, detecting defects that affect the inner surface of the stent in a matter of seconds.
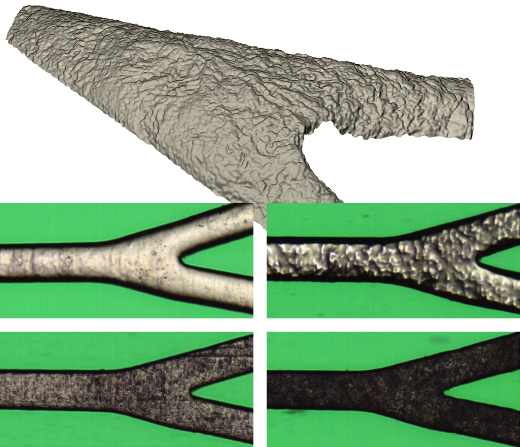

Surface finish
The polishing process of coronary stents is the most critical process in their manufacturing. Guaranteeing a proper surface finish of the devices is essential since the devices will be implanted inside body vessels and their external surface will be in direct contact with the patient arteries.
Surface finish is characterized from 2D unrolled images by the measurement of specific structural parameters calculated in regions of interest. By establishing tolerance values of these parameters, it is possible to set an objective criterion for the surface quality of the stent at selected points. This allows the system to determine and display surface finish results together with the rest of the inspection results.
3D Measurement of defects
The 3D technology embedded in our inspection systems gives the engineers and operators the possibility to perform detailed measurements of the deviations found, providing them with 3D information of any structure found.
This capability allows a detailed characterization of surface defects in the nanometric scale during product development, avoiding the need to characterize the defects by means of destructive tests such as Scanning Electron Microscope.
During stent inspection in production, this feature allows to characterize in few seconds any surface deviation found, providing operators with additional information to take the best decision about the quality of the produced stents.


Drug-eluting stents
In order to keep the artery blockage to coming back after the intervention, some stents are coated with a drug-eluting polymer layer, conforming what are known as drug-eluting stents (DES).
The introduction of such devices to the market brought new challenges to their inspection for Quality Control. Since the dose of drug on each stent needs to be maintained within certain numbers to maximize effectivity, the drug-eluting coating thickness and uniformity are critical manufacturing parameters for this type of stents.
With Sensofar Medical’s 3D technology, the thickness of optically transparent coatings can be mapped along the stents with nanometric resolution in a matter of seconds, allowing for a minimum coating thickness measurement of 2 microns.



